 This is an important technique for analyzing protein-DNA interactions.Īlthough dimethyl sulfate is highly effective and affordable, its toxicity has encouraged the use of other methylating reagents. Nuclease S1 can then be used to cut the DNA in single-stranded regions (anywhere with a methylated adenine). Upon re-annealing, these methyl groups interfere with adenine-guanine base-pairing. Dimethyl sulfate also methylates adenine in single-stranded portions of DNA (e.g., those with proteins like RNA polymerase progressively melting and re-annealing the DNA). These mutations can be detected via sequencing, and the RNA is inferred to be single-stranded at bases with above-background mutation rates.ĭimethyl sulfate can effect the base-specific cleavage of DNA by attacking the imidazole rings present in guanine. Then the RNA is reverse-transcribed the reverse transcriptase frequently adds an incorrect DNA base when it encounters a methylated RNA base. Using the method known as DMS-MaPseq, RNA is incubated with DMS to methylate unpaired bases. At neutral pH, DMS methylates unpaired adenine and cytosine residues at their canonical Watson-Crick faces, but it cannot methylate base-paired nucleotides. RC(O)SH + (CH 3O) 2SO 2 → RC(O)S(CH 3) + HOSO 3CH 3 Reactions with nucleic acids ĭimethyl sulfate (DMS) is used to determine the secondary structure of RNA. This method has been used to prepare thioesters: Similar to the methylation of alcohols, mercaptide salts are easily methylated by Me 2SO 4: RS −Na + + (CH 3O) 2SO 2 → RSCH 3 + Na(CH 3)SO 4Īn example is: p-CH 3C 6H 4SO 2Na + (CH 3O) 2SO 2 → p-CH 3C 6H 4SO 2CH 3 + Na(CH 3)SO 4 Methylation to create a tertiary amine is illustrated as: CH 3(C 6H 4)NH 2 + (CH 3O) 2SO 2 (in NaHCO 3 aq.) → CH 3(C 6H 4)N(CH 3) 2 + Na(CH 3)SO 4 Methylation at sulfur Quaternized fatty ammonium compounds are used as a surfactant or fabric softeners. Me 2SO 4 is used to prepare both quaternary ammonium salts or tertiary amines:Ĭ 6H 5CH=NC 4H 9 + (CH 3O) 2SO 2 → C 6H 5CH=N +(CH 3)C 4H 9 + CH 3OSO 3 − The methylation of ketones is called the Lavergne reaction. The methylation of sugars is called Haworth methylation. 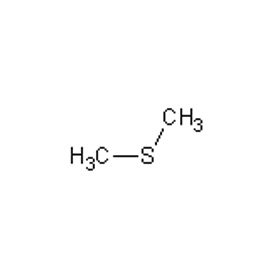
Some simple alcohols are also suitably methylated, as illustrated by the conversion of tert-butanol to t-butyl methyl ether:Ģ (CH 3) 3COH + (CH 3O) 2SO 2 → 2 (CH 3) 3COCH 3 + H 2SO 4Īlkoxide salts are rapidly methylated: RO − Na + + (CH 3O) 2SO 2 → ROCH 3 + Na(CH 3)SO 4 Compared to other methylating agents, dimethyl sulfate is preferred by the industry because of its low cost and high reactivity.Ĭommonly Me 2SO 4 is employed to methylate phenols. Methyl transfer is assumed to occur via an S N2 reaction. One methyl group is transferred more quickly than the second. 
(CH 3) 2O + SO 3 → (CH 3) 2SO 4 Reactions and uses ĭimethyl sulfate is a reagent for the methylation of phenols, amines, and thiols. A common process is the continuous reaction of dimethyl ether with sulfur trioxide. The reaction of methyl nitrite and methyl chlorosulfonate also results in dimethyl sulfate: CH 3ONO + (CH 3)OSO 2Cl → (CH 3) 2SO 4 + NOClĭimethyl sulfate has been produced commercially since the 1920s. Production ĭimethyl sulfate can be synthesized in the laboratory by several methods, the simplest being the esterification of sulfuric acid with methanol: 2 CH 3OH + H 2SO 4 → (CH 3) 2SO 4 + 2 H 2OĪt higher temperatures, dimethyl sulfate decomposes. It was investigated as a candidate for possible use in chemical warfare in World War I in 75% to 25% mixture with methyl chlorosulfonate (CH 3ClO 3S) called "C-stoff" in Germany, or with chlorosulfonic acid called "Rationite" in France. Claesson later extensively studied its preparation. Its use as a laboratory reagent has been superseded to some extent by methyl triflate, CF 3SO 3CH 3, the methyl ester of trifluoromethanesulfonic acid.ĭimethyl sulfate was discovered in the early 19th century in an impure form. Like all strong alkylating agents, Me 2SO 4 is extremely toxic. 
Me 2SO 4 is a colourless oily liquid with a slight onion-like odour (although smelling it would represent significant exposure). Me 2SO 4 is mainly used as a methylating agent in organic synthesis. As the diester of methanol and sulfuric acid, its formula is often written as ( CH 3) 2 SO 4 or Me 2SO 4, where CH 3 or Me is methyl. Dimethyl sulfate ( DMS) is a chemical compound with formula (CH 3O) 2SO 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
